Stem cell therapy for heart failure is an emerging approach in regenerative medicine that focuses on repairing damaged heart tissue. Stem cells are unique cells capable of developing into specialized cells and supporting the body’s natural healing processes. Researchers are studying how stem cells may help improve blood vessel growth, support cardiac tissue repair, and enhance overall heart function in patients with heart failure. Although research is ongoing, stem cell therapy continues to be explored as a potential supportive strategy in cardiovascular regenerative medicine.
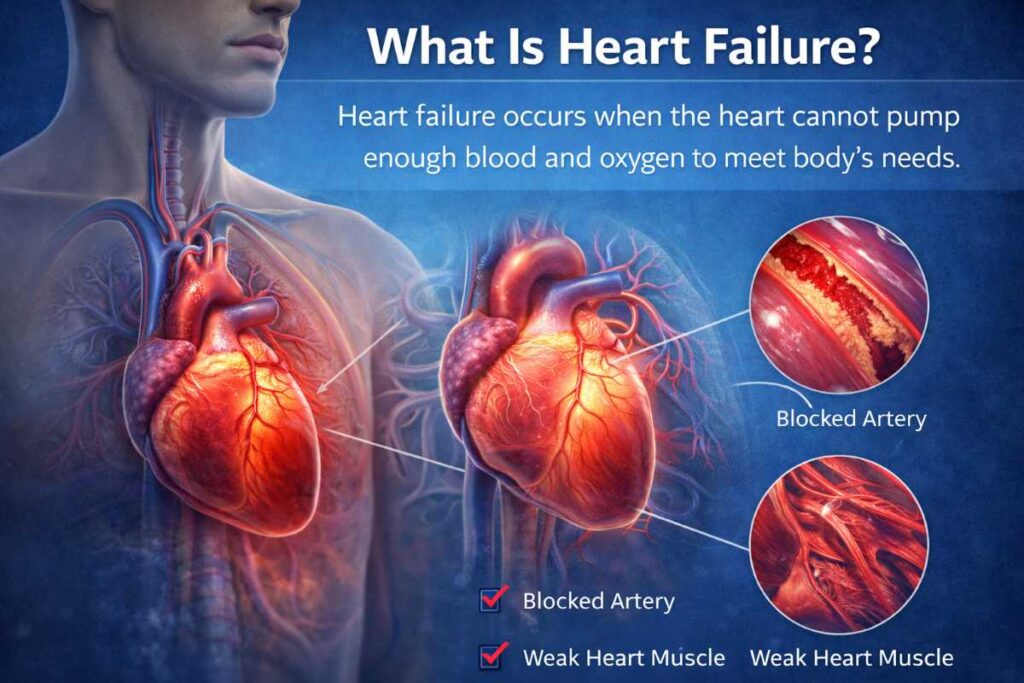
What is Heart Failure?
Heart failure is a condition in which the heart is not able to pump enough blood through the body to maintain normal function. The cause of this can be either functional or structural disorder of the heart. Common causes of heart failure include cardiomyopathy, hypertension, heart valve problems, and coronary artery disease. There is no present cure for heart failure.
Can stem cells help treat heart failure?
CD 34+ stem cells have the ability to not only home into the damaged areas but also to trigger a series of very beneficial biological events which culminate in healing of the heart muscle. For example, animal studies have demonstrated that stem cell therapy will cause new muscle cells to be formed through stimulation of dormant stem cells that are already inside the heart muscle. In these studies, the administered stem cell also transformed into new heart muscle cells. Our technology allows you own body to release large volumes (up to eight times your present level) of CD 34+ stem cells.
Which kinds of stem cells are used to treat heart disease and where do they come from?
The CD 34+ stem cells are ideally suited for the treatment of heart failure. HBOT exposure causes the bone marrow to mobilize cascades of CD 34+ stem cells from the patient’s own body. Naturally, these cells are keyed with the patient’s own DNA, therefore these stem cells pose no rejection risk because the body does not recognize them as foreign.
We offer two protocols to mobilize the CD 34+ stem cells:
Two 60-minute hyperbaric exposures daily for 20 consecutive days (each exposure is for approximately 75-minutes in total duration)
One 90-minute hyperbaric exposure daily for 10 consecutive days (each exposure is approximately 2- hours in total duration)
It is suggested that the patient/caregiver read the University of Pennsylvania School of Medicine “Stem Cell Mobilization Study”. Please click here for copy of full study to understand how Hyperbaric Oxygen Therapy (HBOT) has been proven to mobilize CD 34+ stem cells after 40 cumulative hours of exposure.
The University of Texas School of Medicine is using stem cell technology to grow new heart valves in the lab.
The following extract from a scientific study shows that adult (age appropriate) stem cells are able to offer great hope for patients who present with heart disease.
Challenges in Traditional Stem Cell Mobilization Methods
When we tried to tell the University of Texas Stem Cell Unit (working on growing new heart valves with CD 34+ adult stem cells) about the proven fact that HBOT increases the amount of CD34+ stem cells (keyed with your own DNA) by a factor of 800% they hung up on us!
Clearly, this discovery would have caused them to loose their funding, so they continue to try to find ways to mobilize more stem cells with drugs and undesirable side affects.
Researchers have demonstrated for the first time that injecting adult bone marrow stem cells into skeletal muscle can repair cardiac tissue, reversing heart failure.
Using an animal model, the researchers showed that this non-invasive procedure increased myocytes, or heart cells, by two-fold and reduced cardiac tissue injury by 60 percent.
The therapy also improved function of the left ventricle, the primary pumping chamber of the heart, by 40 percent and reduced fibrosis, the hardening of the heart lining that impairs its ability to contract, by up to 50 percent.
“This work demonstrates a novel non-invasive mesenchymal stem cell therapeutic regimen for heart failure based on an intramuscular delivery route,” said Techung Lee, senior author on the paper.
Mesenchymal stem cells are found in the bone marrow and can differentiate into a variety of cell types.
How Mesenchymal Stem Cells Support Heart Tissue Healing
“Injecting mesenchymal stem cells or factors released by mesenchymal stem cells improved ventricular function, promoted myocardial regeneration, lessened apoptosis (cell death) and fibrotic remodeling, recruited bone marrow progenitor cells and induced myocardial expression of multiple growth factor genes,” Lee said.
“These findings highlight the critical ‘cross-talks’ between the injected mesenchymal stem cells and host tissues, culminating in effective cardiac repair for the failing heart.”
The heart disease death rate has dropped significantly in the last three decades due to better treatments, resulting in large numbers of people living with heart failure. This advance has lead to another health hurdle: The only therapy available to reverse the decline in cardiac function is heart transplantation, and donor hearts are very scarce.
Clinical trials of myocardial stem cell therapy traditionally have relied on surgery, infusing the stem cells directly into the heart or injecting them into the myocardium, the heart muscle, invasive methods that can result in harmful scar tissue, arrhythmia, calcification or small vessel blockages.
Why Non-Invasive Stem Cell Therapy Approaches Are Being Explored
“In our research with a swine model of heart failure,” said Lee, “we’ve found that only 1-to-2 percent of mesenchymal stem cells infused into the myocardium grafted into the heart, and there was no evidence that they differentiated into heart muscle cells. In addition, diseased tissue is not a healthy environment for cell growth.
“For these reasons, and because patients with heart failure are not good surgical risks, it made sense to explore a non-invasive cell delivery approach,” said Lee. “An important feature of mesenchymal stem cells is their ability to produce a plethora of tissue healing effects, known as “trophic factors,” which can be harnessed for stem cell therapy for heart failure.
Lee noted that the multiple trophic factors produced by mesenchymal stem cells have been shown in the literature to be capable of reducing tissue injury, inhibiting fibrosis, promoting angiogenesis, stimulating recruitment and proliferation of tissue stem cells, and reducing inflammatory oxidative stress, a common cause of cardiovascular disease and heart failure.
“Since skeletal muscle is the most abundant tissue in the body and can withstand repeated injection of large number of stem cells, we thought it would be a good method to deliver mesenchymal stem cells,” Lee said. “We hypothesized that mesenchymal stem cells, via secretion of these functionally synergistic trophic factors, would be able to rescue the failing heart even when delivered away from the myocardium.
Future Potential of Mesenchymal Stem Cell Therapy for Heart Failure
“This study proves our hypothesis,” said Lee. “We’ve demonstrated that injecting mesenchymal stem cells, or trophic factors released by mesenchymal stem cells, into skeletal muscle improved ventricular function, promoted regeneration of heart tissue, decreased cell death and improved other factors that cause heart failure.
“This non-invasive stem cell administration regimen, if validated clinically, is expected to facilitate future stem cell therapy for heart failure.”
Lee said the next step is to use genetic and pharmacological engineering to make the stem cells more active, so good therapeutic effects can be achieved with fewer cells.
“That is our goal. It would reduce the cost of stem cell therapy and make it more affordable for patients in the future.”
References:
1. Techung Lee, et al. Heart Failure Therapy Mediated by the Trophic Activities of Bone Marrow Mesenchymal Stem Cells: A Non-invasive Therapeutic Regimen. Am J Physiol Heart Circ Physiol (April 24, 2009). doi:10.1152/ajpheart.00186.2009.
2. University of Pennsylvania School of Medicine “Stem Cell Mobilization Study”..



